COVID-19 vaccine with IPD nanoparticles wins full approval abroad
Message boards : News : COVID-19 vaccine with IPD nanoparticles wins full approval abroad
| Author | Message |
|---|---|
|
Admin Project administrator Send message Joined: 1 Jul 05 Posts: 5146 Credit: 0 RAC: 0 |
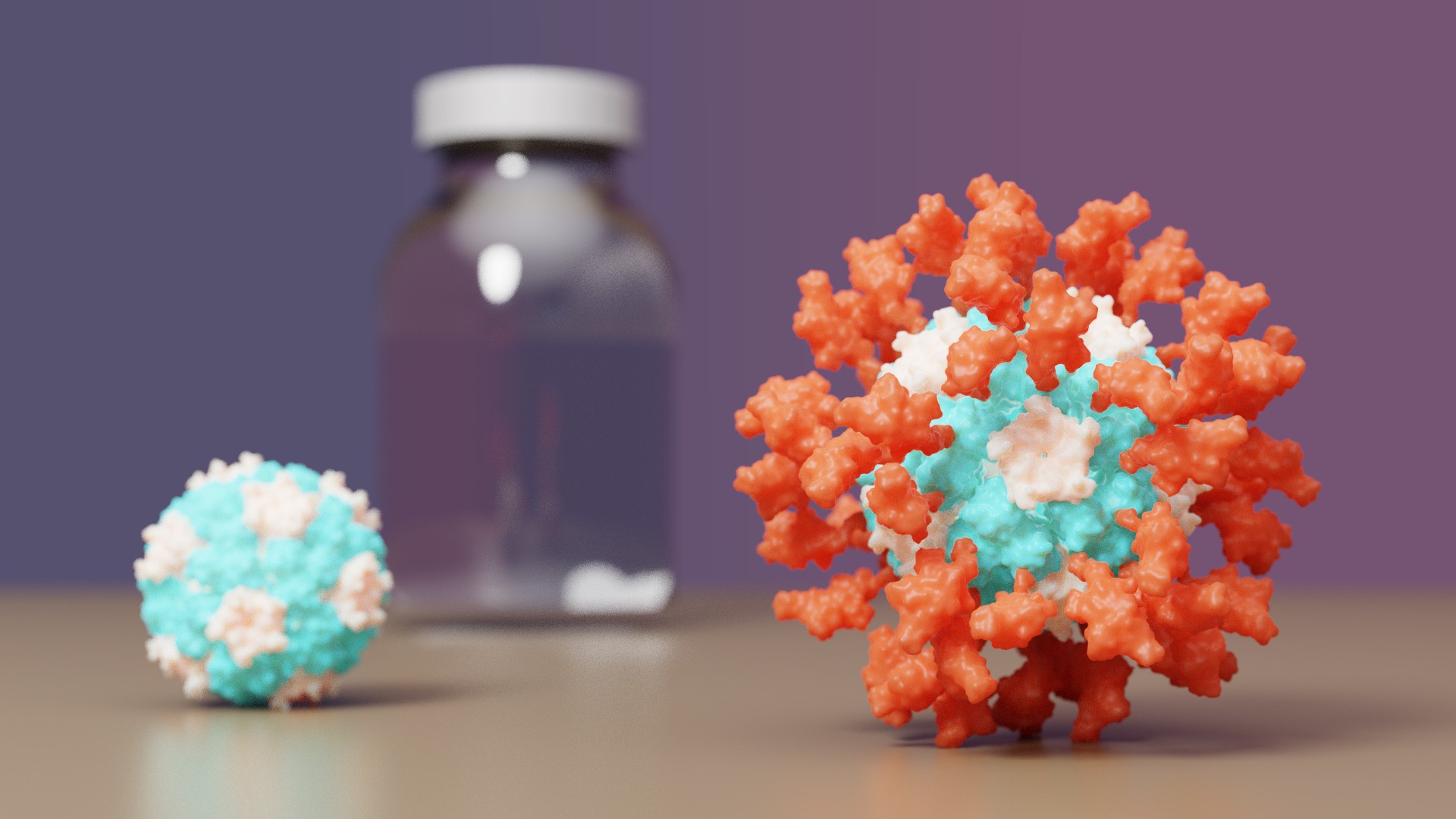 The IPD is excited to announce it's first designed protein medicine with full approval abroad. Congrats and thank you to all Rosetta@home contributors! The computing you have provided has greatly aided in de novo protein design challenges such as vaccine development leading to breakthroughs like this. For more information you can visit the IPD vaccine news post. A video is also available here. From the IPD news site: • Clinical testing found the vaccine outperforms Oxford/AstraZeneca’s • The protein-based vaccine, now called SKYCovione, does not require deep freezing • University of Washington to waive royalty fees for the duration of the pandemic • South Korea to purchase 10 million doses for domestic use A vaccine for COVID-19 developed at the University of Washington School of Medicine has been approved by the Korean Ministry of Food and Drug Safety for use in individuals 18 years of age and older. The vaccine, now known as SKYCovione, was found to be more effective than the Oxford/AstraZeneca vaccine sold under the brand names Covishield and Vaxzevria. SK bioscience, the company leading the SKYCovione’s clinical development abroad, is now seeking approval for its use in the United Kingdom and beyond. If approved by the World Health Organization, the vaccine will be made available through COVAX, an international effort to equitably distribute COVID-19 vaccines around the world. In addition, the South Korean government has agreed to purchase 10 million doses for domestic use. The Seattle scientists behind the new vaccine sought to create a ‘second-generation’ COVID-19 vaccine that is safe, effective at low doses, simple to manufacture, and stable without deep freezing. These attributes could enable vaccination at a global scale by reaching people in areas where medical, transportation, and storage resources are limited. “We know more than two billion people worldwide have not received a single dose of vaccine,” said David Veesler, associate professor of biochemistry at UW School of Medicine and co-developer of the vaccine. “If our vaccine is distributed through COVAX, it will allow it to reach people who need access.” The University of Washington is licensing the vaccine technology royalty-free for the duration of the pandemic. Congrats and thank you again to all R@h contributors! |
|
.clair. Send message Joined: 2 Jan 07 Posts: 274 Credit: 26,399,595 RAC: 0 |
Nice to know it is working I wonder if this was through Python tasks or Rosetta 4.2 |
|
Sid Celery Send message Joined: 11 Feb 08 Posts: 2582 Credit: 47,220,881 RAC: 426 |
Nice to know it is working The first paper in the development of this vaccine was sent in August 2020, revised in Oct 20 and published Oct 31 2020 at Cell all of which is before the results of CASP were published, so there was no Alphafold, Rosettafold nor VBox versions of Rosetta to the best of my memory, unless someone corrects me. The way I remember things going, all the developments in Rosetta were rolled up and released end March/early April 2020 for us to work on and whatever they asked us to process would reasonably have contributed to this outcome. I assume we all received an email from UW with this news on 30th June, to which I replied requesting a post to this forum. I don't know whether this post responds to my request, but I'm very glad they did so Edit: the email received may only have gone out to people who've made donations to UW rather than all Rosetta users  
|
|
Falconet Send message Joined: 9 Mar 09 Posts: 355 Credit: 1,669,337 RAC: 0 |
A month-old article about the need for an additional safety review of the vaccine. "According to the minutes, members of the central pharmaceutical review panel said SKYCovione’s safety was within the permissible level. However, due to the small number of cases, panelists said it was difficult to conclude the safety and that the vaccine needed continued monitoring." |
|
Ostrailly Send message Joined: 1 Dec 22 Posts: 4 Credit: 0 RAC: 0 |
nicesu |
|
AlanG7Mc Send message Joined: 26 Jun 06 Posts: 4 Credit: 884,099 RAC: 0 |
TERMINATION NOTICE: With the stupidly 'soon' deadlines that >You Choose< ... With the way that >I Choose< to run my computer (including >My Choosing< hours-per-day to run it) ... I find that the >Very Last< Unit I will every try for You, cannot complete within deadline. With Your Right and privilege to chose too-soon dead-lines ... With >My Right and Privilege< to run my computer My Way, and to >Volunteer< My Way ... Result: YOU have >Chosen< to Steal 30+ hours of CPU time from another project who actually wants results. And you have just thrown away a Volunteer (Me) who has been here for Seventeen Years. |
|
Sid Celery Send message Joined: 11 Feb 08 Posts: 2582 Credit: 47,220,881 RAC: 426 |
A month-old article about the need for an additional safety review of the vaccine. It looked like this SkyCovione vaccine was going to be dead in the water, but the UK Medicines and Healthcare products Regulatory Agency (MHRA) has just given it full approval (26th May 2023) SKYCovion COVID-19 vaccine authorised by MHRA  
|
Message boards :
News :
COVID-19 vaccine with IPD nanoparticles wins full approval abroad

©2026 University of Washington
https://www.bakerlab.org